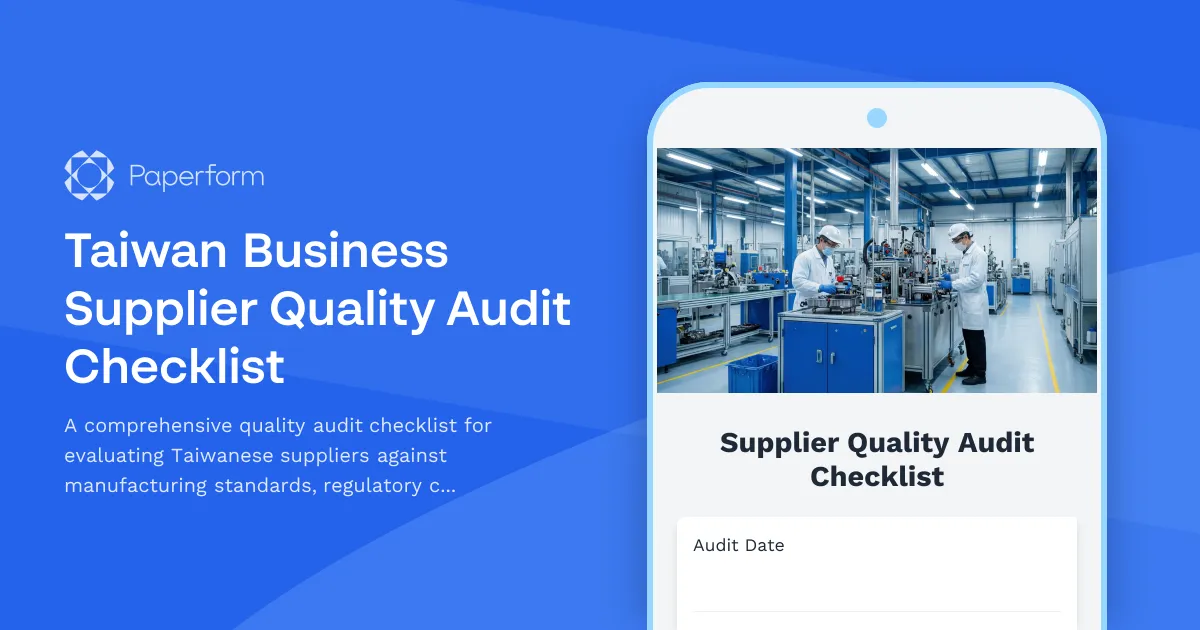
Managing supplier quality in Taiwan's competitive manufacturing sector requires systematic audits that balance regulatory compliance with operational excellence. This Taiwan Business Supplier Quality Audit Checklist helps procurement teams, quality managers, and supply chain professionals conduct thorough supplier evaluations that meet both local regulatory requirements and international quality standards.
Whether you're qualifying new suppliers, conducting annual audits, or investigating quality issues, this template covers critical areas including manufacturing capabilities, quality management systems, regulatory compliance (including Unified Business Number verification), workplace safety, environmental standards, and corrective action planning.
Built for manufacturers, importers, and businesses working with Taiwanese suppliers, this checklist streamlines the audit process from initial documentation review through final scoring and corrective action tracking. The structured format ensures consistency across multiple supplier evaluations while capturing the detailed findings needed for informed sourcing decisions.
Paperform makes it simple to conduct these audits digitally, whether on-site with mobile devices or remotely via shared links. Conditional logic adapts the form based on audit findings, calculations automatically generate compliance scores, and responses flow directly into your quality management system or procurement tools.
For teams managing complex supplier onboarding workflows, Stepper can automate the entire supplier qualification process—triggering audit scheduling, routing corrective action requests to suppliers, sending follow-up reminders, and updating your ERP or supplier database when audits are completed and approved.
More templates like this

BOC Import Entry and Internal Revenue Declaration (IEIRD) Form
Official Philippine BOC form for import entry and internal revenue declaration with tariff classification and duties computation for customs clearance.

Buy American Act Certificate of Compliance
A compliance certificate for contractors to certify that products and materials supplied under government contracts meet Buy American Act domestic content requirements.
Child Labour Prohibition Compliance Certificate
Verify compliance with India's Child Labour Act through age verification, hazardous process exclusion checks, and comprehensive inspection readiness documentation.

Dutch Quality Mark Certification Application Form
A comprehensive certification application form for businesses seeking Dutch quality mark certification, featuring sector-specific standards assessment and audit scheduling functionality.

FDA Certificate of Product Registration (CPR) Renewal Application Form
A comprehensive form for renewing FDA Certificate of Product Registration (CPR) in the Philippines with updated product formulation and labeling information.
FDA Food Facility Registration Renewal Form
Streamline your biennial FDA food facility registration renewal with this comprehensive form. Update facility information, confirm contact details, and designate preferred mailing addresses to maintain compliance with FDA regulations.

French Carbon Footprint Declaration (Bilan Carbone)
A comprehensive French carbon footprint declaration form (Bilan Carbone) for regulatory reporting, featuring emissions calculations, reduction plans, and compliance with French environmental regulations.

ISO Quality Management Certification Application Form (Germany)
A comprehensive application form for German businesses seeking ISO quality management certification, including process documentation, audit scheduling, and accreditation body selection.

Japanese Subcontracting Law (下請法) Compliance Form
Comprehensive compliance form for Japan's Subcontracting Act (Act on the Prevention of Delay in Payment of Subcontract Proceeds, Etc.) ensuring proper payment terms, delivery schedules, and adherence to prohibited practices.

NAFDAC Product Registration Application Form
Complete NAFDAC product registration form for food, drugs, cosmetics, and medical devices in Nigeria. Streamline your regulatory compliance with detailed sections for product formulation, manufacturing details, and laboratory test results.

Taiwan Manufacturing Quality Certification (ISO) Application
A comprehensive ISO certification application form for Taiwanese manufacturers, including company details, process documentation, and audit scheduling.

Taiwan Product Recall Notification Form
A comprehensive notification form for Taiwan FDA product recalls, documenting affected batches, business details, and corrective action plans in compliance with Taiwanese regulatory requirements.


