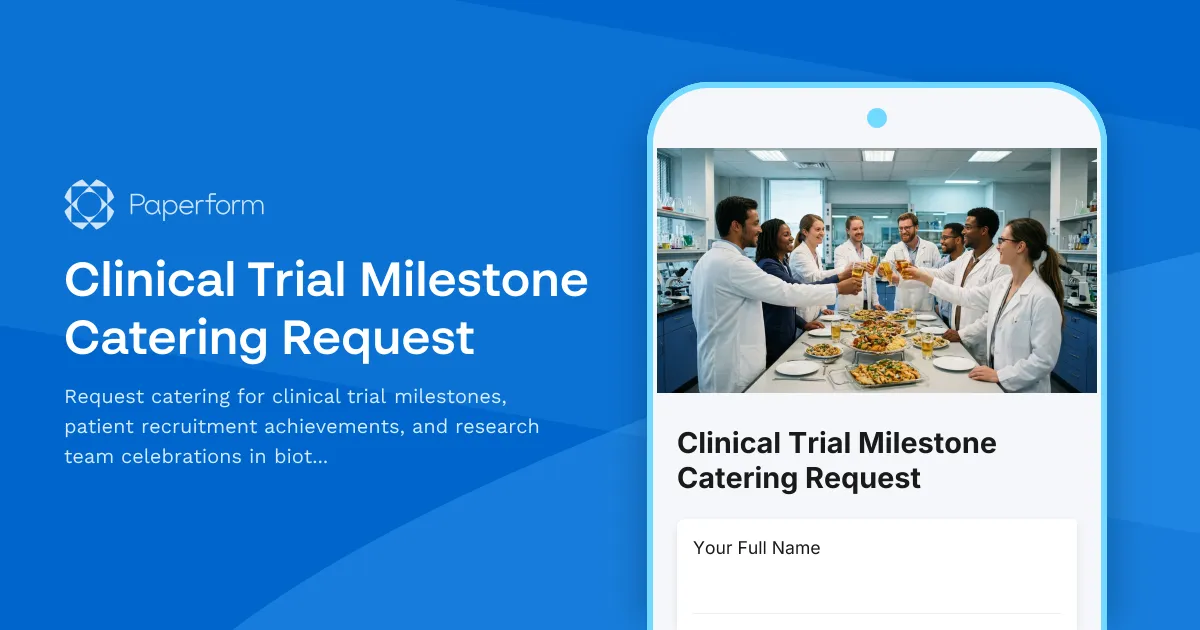
Celebrate Clinical Trial Milestones with Professional Catering
Clinical trial milestones deserve recognition. Whether you're celebrating successful patient enrollment, study launch events, or research team achievements, coordinating catering for your biotech or pharmaceutical team shouldn't add to your workload.
This Clinical Trial Milestone Catering Request Form streamlines food ordering for research coordinators, clinical operations teams, and trial site managers. Designed specifically for the biotech and pharmaceutical industry, it captures dietary requirements, headcount projections, budget parameters, and milestone-specific details in one organized submission.
Why Clinical Research Teams Choose This Template
Gene therapy trials, oncology studies, and multi-site clinical research programs operate on tight timelines with rigorous compliance standards. When you're managing patient recruitment targets, IRB submissions, and protocol amendments, the last thing you need is a complicated catering request process.
This form template helps clinical research teams:
- Coordinate milestone celebrations for first patient enrolled, target enrollment achieved, or trial launch events
- Capture dietary restrictions and allergies to ensure safe, inclusive meals for research staff and stakeholders
- Plan within budget constraints whether you're ordering lunch for 15 coordinators or hosting a 100-person site initiation visit
- Streamline internal approvals with clear visibility into event purpose, attendee list, and cost estimates
- Support remote and hybrid teams with delivery coordination across multiple trial sites or home offices
Built for Biotech Workflows
Clinical trial catering involves unique considerations. Research coordinators juggle patient visits, regulatory documentation, and sponsor communications—this form adapts to their reality. You can specify whether the event coincides with investigator meetings, patient recruitment drives, or regulatory milestone achievements.
The form captures essential details like site location, preferred vendors (for facilities with approved caterers), delivery timing around clinical visits, and special requirements for GLP/GCP-compliant facilities with security protocols.
Automate What Comes Next with Stepper
Once a catering request is submitted, there's still work to do: routing for budget approval, notifying facility coordinators, confirming with vendors, and updating the team. That's where Stepper transforms this form from a simple request into an automated workflow.
With Stepper connected to your Paperform, you can automatically:
- Route high-value requests to finance for budget approval
- Send confirmation emails to event organizers with next steps
- Create calendar invites for the catering delivery and event
- Update your shared catering tracker in Airtable, Google Sheets, or Monday.com
- Notify facilities teams about site access requirements
- Track spending against departmental catering budgets
For multi-site clinical trials, you can build conditional workflows that route requests to the appropriate regional operations manager or site administrator based on location. No more chasing approvals through email threads.
Designed for Research Teams, Not Just Forms
Paperform's document-style editor means you can customize this template to match your organization's specific protocols. Add your company logo, embed images from past celebrations, include links to preferred vendor menus, or add conditional logic to show different options for investigator meetings versus internal team events.
Whether you're a CRO managing dozens of trial sites, a biotech startup celebrating your first IND submission, or a university research center hosting NIH site visits, this template adapts to your operational needs. The clean, professional design works equally well on desktop for study coordinators or mobile for principal investigators submitting requests between patient appointments.
Security and Compliance You Can Trust
Clinical research demands data security, and Paperform delivers. With SOC 2 Type II compliance, role-based permissions, and data residency controls, you can trust that your catering requests (which often include attendee names, site locations, and budget information) are handled with enterprise-grade security.
For teams using Paperform across multiple departments or trial programs, Agency+ features let you manage separate forms for each therapeutic area or research site while maintaining centralized oversight and reporting.
Get Started in Minutes
This template comes ready to use with smart defaults for common clinical trial catering scenarios. Simply customize the vendor options, adjust budget thresholds, and connect your preferred integrations through Stepper, Zapier, or Paperform's native integrations with Slack, Google Workspace, and Microsoft 365.
Over 500,000 teams worldwide trust Paperform to handle their most important workflows—from patient intake forms to regulatory submissions. Join research coordinators, clinical operations managers, and biotech teams who've replaced messy email chains and scattered spreadsheets with one flexible platform.
Ready to streamline your clinical trial catering requests? Start with this template and experience how Paperform turns everyday operational tasks into smooth, automated workflows that let your research team focus on advancing science, not chasing lunch orders.
More templates like this
Biotech Patent Filing Celebration Catering Request
Coordinate catering for your biotech patent application celebration with legal team coordination, inventor dietary preferences, and IP milestone recognition planning.
Lab Scientist Appreciation & Milestone Celebration Catering Request
A specialized catering request form for biotech labs to celebrate research milestones, support teams during grant deadlines, and coordinate scientist appreciation meals with dietary accommodations and lab-safe delivery options.
Biotech Lab All-Hands Town Hall Catering Request
A specialized catering request form for biotech laboratories organizing all-hands town hall meetings, accommodating research milestone celebrations and scientific staff dietary needs.
Biotech Lab Team Lunch Coordination Form
Coordinate team lunches for biotech lab staff with cleanroom facility restrictions, allergen-free preparation requirements, and experiment scheduling considerations.

Biotech Laboratory Equipment Validation Party Catering Request
A specialized catering request form for biotech labs celebrating equipment validation milestones, instrument qualification completions, and research capacity expansion events.
Biotech Lab Office Desk Assignment Form
A specialized desk assignment form for biotech labs that manages hot desking, research team clustering, equipment booking, and compliance documentation workspace needs.
Biotech Laboratory Safety Certification Training Catering Request
Request catering for biotech lab safety training, compliance workshops, and milestone celebrations. Coordinate dietary requirements and schedule food service for EHS events.

Biotech Manufacturing Facility Validation Celebration Catering Request
Request catering for biotech facility validation celebrations, FDA approvals, and quality milestone events with team dietary preferences and regulatory-themed party planning.
Biotech Manufacturing Scale-Up Success Celebration Catering Form
A professional catering request form for biotech manufacturing teams celebrating production milestones, scale-up achievements, and commercial launch events with detailed dietary preferences and venue logistics.

Biotech Regulatory Submission Celebration Catering Request Form
Request catering for your regulatory submission celebration. Perfect for biotech teams celebrating FDA filings, quality assurance milestones, and approval anticipation gatherings.
Cryonics Facility Tour Registration Form
Register for a cryonics facility tour with safety briefing, ethical considerations, and strict photography policy acknowledgment. Secure your spot for an educational visit to our preservation facility.
Laboratory Equipment Relocation Request Form
A comprehensive form for requesting the relocation of laboratory and specialized equipment, including calibration requirements, environmental controls, certified handler needs, and insurance details.


