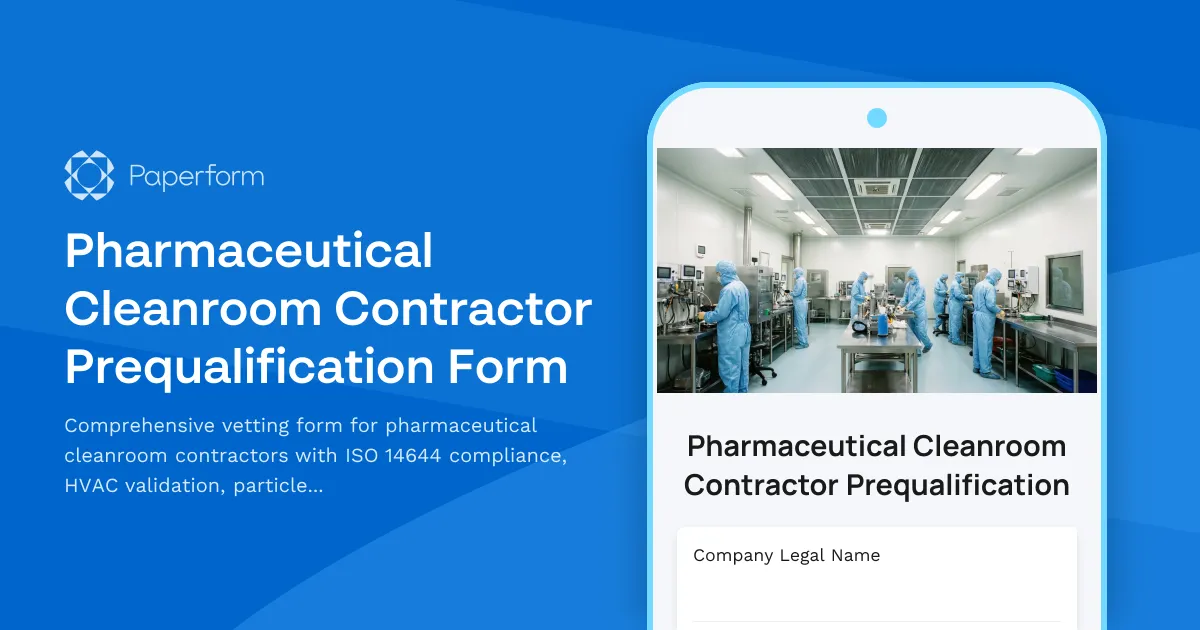
Streamline Your Pharmaceutical Cleanroom Contractor Vetting
When it comes to pharmaceutical cleanroom construction and maintenance, contractor qualification isn't just a formality—it's a critical quality assurance step that directly impacts product safety and regulatory compliance. This Pharmaceutical Cleanroom Contractor Prequalification Form provides a structured approach to evaluating contractors against ISO 14644 standards, HVAC validation requirements, and GMP documentation protocols.
Built specifically for pharmaceutical manufacturers, quality assurance teams, and facilities managers, this template helps you systematically assess contractor capabilities across cleanroom classification expertise, particle counting procedures, environmental monitoring, and validation documentation. The form captures essential certifications, project experience with specific ISO cleanroom classes, HVAC system validation proficiency, and understanding of FDA and EU GMP requirements.
With Paperform's conditional logic, you can tailor follow-up questions based on contractor specializations—whether they focus on cleanroom construction, HVAC installation, certification testing, or ongoing maintenance. The built-in calculation and scoring features help you objectively evaluate and compare multiple contractors, while file upload fields make it easy to collect certifications, insurance documents, and past validation reports in one centralized location.
Once submissions come in, connect this form to your procurement workflow using Stepper to automatically route high-scoring contractors to your technical review team, trigger calendar invites for site visits, update your vendor management system, and send follow-up requests for additional documentation—all without manual data entry.
For pharmaceutical companies managing multiple facilities or qualification processes, Paperform's Agency+ plan lets you maintain separate contractor databases while standardizing your vetting criteria across locations. Plus, with SOC 2 Type II compliance and robust data security, you can confidently collect sensitive vendor information knowing it meets the same high standards you apply to your manufacturing operations.
More templates like this

Laboratory Contractor Pre-Qualification Form
A comprehensive pre-qualification form for specialized laboratory contractors with expertise in cleanroom construction, fume hoods, gas systems, and contamination control.

Radiation Shielding Contractor Pre-Qualification Form
Comprehensive vetting form for radiation shielding contractors specializing in lead lining installation, medical imaging room design, nuclear medicine facilities, and dosimetry verification services.

Medical Device Complaint Escalation Form
Professional complaint escalation form for medical device manufacturers to assess adverse events, FDA MDR reporting requirements, and potential product recall scenarios with comprehensive severity tracking.

Pharmaceutical Vendor Registration Form
A comprehensive pharmaceutical vendor registration form for FDA-compliant suppliers with controlled substance handling, cold chain logistics, and regulatory documentation requirements.
Pharmaceutical Warehouse Visitor Registration Form
A comprehensive visitor registration form for pharmaceutical warehouses with temperature-controlled area access protocols, contamination prevention measures, and full chain of custody documentation for regulatory compliance.
Vendor & Contractor Misconduct Report Form
Report misconduct, code of conduct violations, or contract compliance issues involving external vendors and contractors who interact with your organization.

Blast-Resistant Construction Contractor Qualification Form
A comprehensive prequalification form for contractors specializing in blast-resistant construction, covering explosive engineering capabilities, reinforced concrete design expertise, blast door installation, and safety testing protocols.

Blood Pressure Monitor Contractor Pre-Qualification Form
Pre-qualify manufacturers and contractors for blood pressure monitor production, including cuff assembly, calibration equipment, display testing, and carrying case manufacturing capabilities.
Controlled Environment Testing Vendor Assessment Form
A comprehensive vendor evaluation form designed for pharmaceutical facilities to assess controlled environment testing vendors, including validation protocols, documentation standards, regulatory compliance, and requalification scheduling.

Crash Cart Contractor Pre-Qualification Form
Pre-qualify specialized contractors for crash cart manufacturing, including drawer assembly, electrical systems, accessory mounting, and powder coating capabilities.

Danish Hazardous Materials Handling Permit Application
A comprehensive application form for Danish businesses to apply for hazardous materials handling permits, including CVR registration, safety documentation, and compliance with Danish environmental regulations.

FDA Adverse Event Reporting Form for Medical Device Manufacturers
Streamlined FDA adverse event reporting form for medical device manufacturers to document device-related incidents, patient outcomes, and maintain regulatory compliance with timeline tracking.


